Living with COVID-19: How a ‘game-changing’ pill might help us manage the pandemic
READ MORE
Living with COVID-19
The coronavirus isn’t going away for good, but an oral treatment could be just around the corner. For the second time since the start of the pandemic, the coronavirus is on the retreat again after the delta variant surged in the summer. Preliminary data suggests Merck’s pill could reduce the chance of hospitalization by 50%. Could it be found in pharmacies sometime soon? This is The N&O’s special report.
Expand All
Living with COVID-19: How a ‘game-changing’ pill might help us manage the pandemic
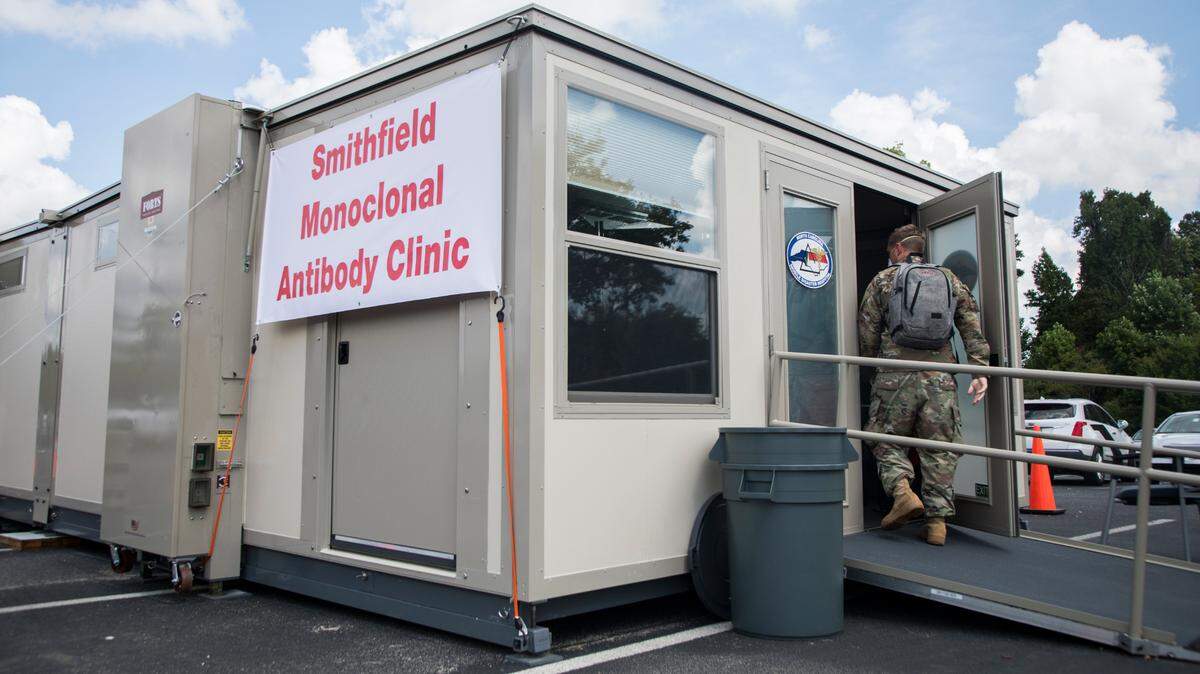
There is still no cure for COVID, but there are treatment options. What’s available in NC?
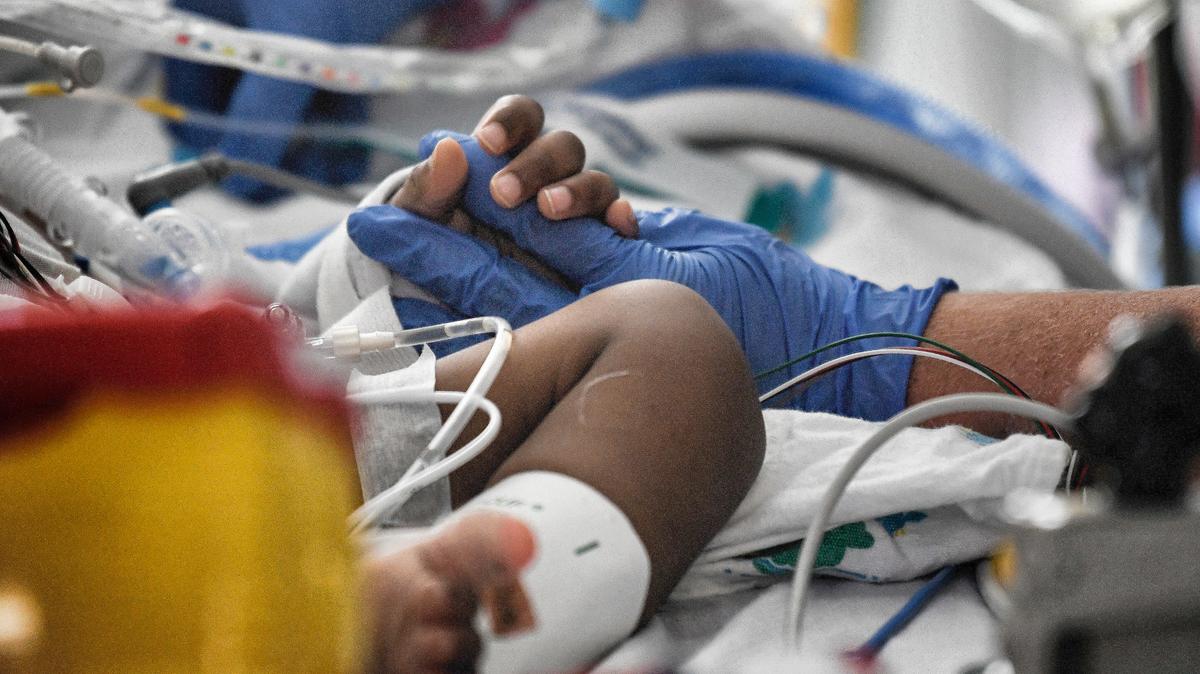
COVID-19 treatments are not vaccine replacements. Here’s why
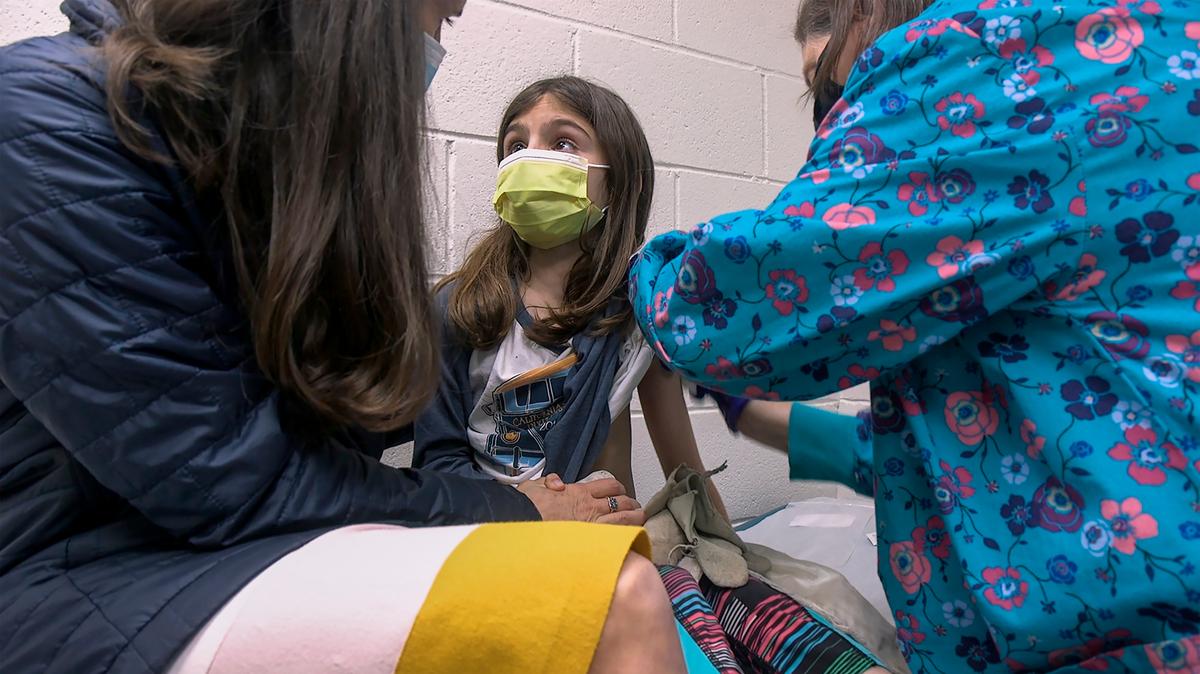
Safe for kids? Duke doctor answers questions about COVID vaccines for children 5 to 11
See which Triangle counties and towns are keeping mask mandates in place, and why
For the second time since COVID-19 was first reported in North Carolina in early 2020, the deadly respiratory disease is on the retreat again after the delta variant surged in the summer.
But the decline probably won’t last as scientists say COVID-19 is likely to be with us for good in some form or another in the years ahead.
“I think we’re going to have to accept that we’re living with it to some extent. It’s clearly not going away,” said Dr. Cameron Wolfe, an infectious disease expert at Duke University. “Adjusting to co-exist with COVID is the way we need to think about this.”
But the first effective treatment available at a local pharmacy could be coming soon.
In October the pharmaceutical company Merck, based in part from research done at the University of North Carolina at Chapel Hill, submitted an application to the Food and Drug Administration asking it to authorize the first antiviral pill to treat COVID-19.
This drug, known as molnupiravir, could reduce a COVID-positve person’s chances of being hospitalized by half, just by taking a pill for a few days, the company said.
Full FDA authorization is expected by the end of the year.
‘The proverbial game-changer’
Since COVID vaccines became available last December, more than half of North Carolinians have taken their shots.
And the Centers for Disease Control and Prevention on Tuesday endorsed the Pfizer-BioNTech vaccine for children age 5 to 11, making an additional 890,000 people in North Carolina eligible. Those shots could start becoming available as soon as Wednesday.
But 44% of state’s population, nearly 4.7 million people, were still not fully vaccinated, as of Monday.
During the summer’s delta bump in cases, the COVID-19 patients who helped push hospital beds and intensive care units to capacity were overwhelmingly unvaccinated.
Dr. David Wohl, an infectious disease expert at UNC-Chapel Hill who helped research the antiviral pill during trials, said Merck’s oral drug could prevent future hospitalizations in another wave.
He called the new treatment “the proverbial game-changer.”
“The fear that we now have when any of us gets diagnosed with COVID-19 is that you’re going to lose the COVID lottery,” Wohl said. “If people with COVID-19 don’t end up in the hospital or don’t end up in the ICU, then we’re in a different world than we are right now. Then it’s more like a cold than a deadly pandemic.”
The Merck pill would not be the only treatment for COVID-19. The FDA authorized use of monoclonal antibody therapies for COVID-19 a year ago, but they must be administered in a clinic or physician’s office through an IV or a shot. Arranging those treatments quickly can be difficult in countries like the United States and impossible in less developed parts of the world.
Merck’s antiviral pill would be available by prescription at a pharmacy and taken twice daily for five days. That ease of use would increase accessibility, Wohl said.
“That would just cast a wider net of protection,” Wohl said.
UNC’s role
Molnupiravir was first developed at Emory University in Atlanta in 2014. Researchers there, seeing the drug’s potential, contacted other universities, including UNC, to help study the drug’s benefits.
UNC researchers began work on molnupiravir in 2016 and found it could be used against coronaviruses. Among these viruses were MERS and SARS-CoV, a virus similar to SARS-CoV-2, the virus that causes COVID-19.
SARS-CoV caused an outbreak of Severe Acute Respiratory Syndrome, a disease that infected over 8,000 people worldwide from 2002 to 2004, according to the CDC.
Dr. William Fischer, director of emerging pathogens at the UNC Institute for Global Health and Infectious Diseases, said the work at UNC to identify molnupiravir’s use against coronaviruses such as COVID-19 was led by researchers Tim Sheehan and Ralph Baric years before the pandemic hit.
“It speaks really highly to how strategic Ralph had been in trying to identify promising therapeutics for coronaviruses,” said Fischer, who also led clinical trials of Merck’s drug. “This was really part of Ralph’s attempt to try to identify therapeutics before they were needed, which is exactly the right strategy.”
Can molnupiravir end the pandemic?
The SARS outbreak was contained in 2003; there haven’t been any known cases since 2004. Health experts say that’s in part because the SARS virus was so deadly — people were less likely to unknowingly spread it to others.
COVID-19 is much more contagious. While deadly, particularly among older people and those with weakened immune systems, the virus is easily transmitted. It’s not likely to disappear, even with vaccines and treatments like molnupiravir and monoclonal antibodies, health experts say.
Even if developed countries are able to control the virus through vaccination alone, it’s likely the virus will live and spread in countries with lower vaccination rates. Fewer than 9% of people living on the continent of Africa are vaccinated, in part because the U.S. and Europe have been slow to share the vaccines, The New York Times reported.
“This is a really dangerous strategy of just focusing on the U.S. or Europe or high-income countries,” Fischer said. “These nationalistic approaches are really dangerous, because we allow the virus to continue to replicate.”
Merck has made an agreement to allow molnupiravir to be produced and sold in developing countries, making the treatment potentially more accessible than vaccines in certain places.
But health experts say the drug isn’t a substitute for vaccination.
Preliminary data suggests Merck’s pill could reduce the chance of hospitalization by 50%. But a September study from the CDC found that the vaccines from Moderna, Pfizer and Johnson & Johnson were respectively 93%, 88% and 71% effective in preventing hospitalization.
“If you’re using this or using any oral treatment, for that matter … and assume that’s a way to get away from getting vaccinated, you are fundamentally misunderstanding the degree to which it protects you from hospitalization,” Wolfe said. “They are not that effective. If I could give a person one vaccine or give them a course of molnupiravir, I would give them the vaccine in a heartbeat.”
And the more people remain unvaccinated globally, the more the virus has the chance to replicate and spread to other parts of the world.
“No matter how well the U.S. does, we are always a little bit exposed now to what’s happening with COVID elsewhere,” Wolfe said.
The combination of vaccines and treatments could render COVID-19 as manageable as seasonal influenza. COVID-19 has killed about 750,000 people in the U.S. since the start of 2020. The less deadly flu normally helps fill hospitals each winter and kills tens of thousands of Americans a year, a toll we’ve come to tolerate, Wolfe said.
He hopes the country will do better with the coronavirus.
“There’s no realistic expectation that this goes away,” he said. “The realistic expectation is that it becomes a relatively uncommon illness that may have some seasonality to it and may still be a much more significant problem in the immunosuppressed or the obese or elderly or unvaccinated in particular but that ticks on quietly in the background.”
This story was originally published November 3, 2021 at 6:00 AM with the headline "Living with COVID-19: How a ‘game-changing’ pill might help us manage the pandemic."
